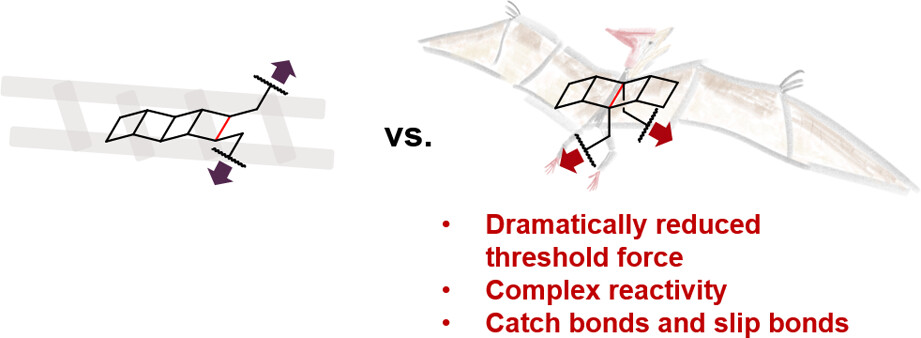
Pterodactylane is a [4]-ladderane with substituents on the central rung. Comparing the mechanochemistry of the [4]-ladderane structure when pulled from the central rung versus the end rung revealed a striking difference in the threshold force of mechanoactivation: the threshold force is dramatically lowered from 1.9 nN when pulled on the end rung to 0.7 nN when pulled on the central rung. We investigated the bicyclic products formed from the mechanochemical activation of pterodactylane experimentally and computationally, which are distinct from the mechanochemical products of ladderanes being activated from the end rung. We compared the products of pterodactylane’s mechanochemical and thermal activation to reveal differences and similarities in the mechanochemical and thermal pathways of pterodactylane transformation. Interestingly, we also discovered the presence of elementary steps that are accelerated or suppressed by force within the same mechanochemical reaction of pterodactylane, suggesting rich mechanochemical manifolds of multicyclic structures. We rationalized the greatly enhanced mechanochemical reactivity of the central rung of pterodactylane and discovered force-free ground state bond length to be a good low-cost predictor of the threshold force for cyclobutane-based mechanophores. These findings advance our understanding of mechanochemical reactivities and pathways, and they will guide future designs of mechanophores with low threshold forces to facilitate their applications in force-responsive materials.
Read more in:
Efficient Acceleration of Reaction Discovery in the Ab Initio Nanoreactor: Phenyl Radical Oxidation Chemistry
Matías Horst , Jan Meisner, Jinghui Yang, Tatiana B. Kouznetsova, Stephen L. Craig, Todd J. Martínez, Yan Xia
J. Am. Chem. Soc., 146, 1, 884-891, 2023